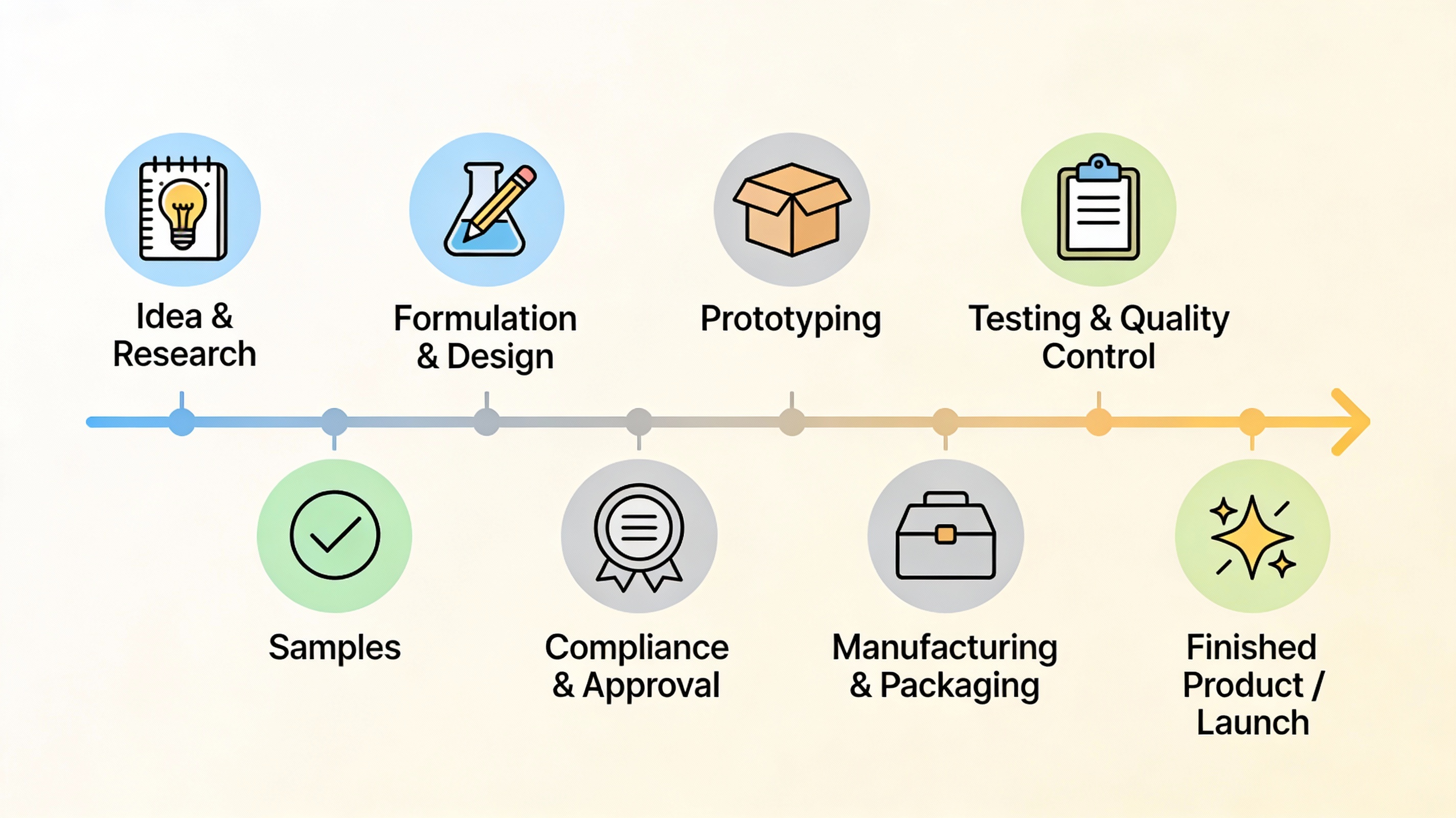
The Reality of Importing Wellness Products from India
India produces some of the world's most sought-after Ayurvedic ingredients and formulations, but the import process can feel opaque if you don't know what to expect. The good news: with the right partner and clear documentation, it's straightforward.
Step 1: Understanding What You're Actually Importing
Product Classification Matters
Ayurvedic products fall into different regulatory categories depending on your destination market:
Dietary Supplements (US, Canada, Australia)
- Regulated as food products
- Requires ingredient compliance with approved lists
- Structure/function claims allowed (but not disease claims)
- FDA registration for facilities (US market)
Traditional Herbal Medicinal Products (EU/UK)
- May require Traditional Herbal Registration (THR)
- Stricter documentation requirements
- Need evidence of traditional use (30+ years, including 15+ in EU)
- Higher regulatory barrier but premium market positioning
Cosmetics (if topical wellness products)
- Different safety assessment requirements
- Specific labeling regulations
- Prohibited ingredients lists vary by region
Why this matters: Your supplier needs to formulate and document products according to your target market's classification.
Step 2: Essential Documentation Checklist
From Your Supplier (Minimum Requirements):
✓ Certificate of Analysis (CoA)
- Confirms product specifications
- Shows test results for purity, potency
- Includes batch/lot numbers
- Must be from accredited laboratory
✓ GMP Certificate
- Good Manufacturing Practice certification
- Shows facility meets quality standards
- Should be from recognized certifying body
- Updated annually
✓ Phytosanitary Certificate (for herb-based products)
- Confirms products free from pests/diseases
- Required by agricultural departments
- Essential for customs clearance
✓ Certificate of Origin
- Declares country of manufacture
- May affect duty rates
- Required for customs valuation
✓ Material Safety Data Sheet (MSDS)
- Safety information for handling/transport
- Required for shipping certain ingredients
- Protects workers and end users
✓ Heavy Metal Testing Reports
- Tests for lead, arsenic, mercury, cadmium
- Critical for herbal products
- Must meet destination country limits
✓ Microbial Testing
- Total plate count
- Absence of E. coli, Salmonella, pathogens
- Essential for ingestible products
For Your Market (You'll Need):
✓ Import License (if required in your country)
✓ Product Registration (for regulated markets like UAE, Singapore)
✓ Customs Broker (recommended for first-time importers)
✓ Insurance Certificate (marine cargo insurance)
Step 3: Regulatory Compliance by Market
United States
- FDA doesn't pre-approve dietary supplements
- Manufacturers must follow cGMP regulations
- Products can't make disease claims
- Facility registration required for importers
- Timeline: First shipment can clear in 3-7 days with proper documentation
European Union
- Traditional Herbal Registration (THR) for medicinal claims
- Novel Food assessment for certain ingredients
- Stricter labeling requirements
- Must appoint EU Responsible Person
- Timeline: Full THR can take 18-24 months; simpler routes available for food supplements
United Kingdom (Post-Brexit)
- Similar to EU but separate registration
- MHRA oversight for herbal medicines
- Food supplements route is faster
- Timeline: 2-4 weeks for compliant shipments
Canada
- Natural Product Number (NPN) or Homeopathic Medicine Number required
- Site license for importers
- Pre-market approval process
- Timeline: NPN application 6-12 months
Australia
- TGA regulation (Therapeutic Goods Administration)
- Products need AUST L (listed) or AUST R (registered)
- Sponsor license required
- Timeline: AUST L listing 4-6 months
Step 4: The Shipping Process Demystified
Typical Timeline from Order to Delivery:
Week 1-2: Production & Documentation
- Manufacturing of your order
- Quality testing and certification
- Document preparation
Week 3: Pre-Shipment
- Customs documentation filed
- Shipping arranged
- Insurance secured
Week 4-6: Transit
- Sea freight: 4-6 weeks (most cost-effective)
- Air freight: 5-7 days (for urgent orders)
- Express courier: 3-5 days (small quantities)
Week 7: Customs Clearance
- Document review by customs
- Possible inspection (random or first-time importer)
- Duty and tax payment
- Release to your warehouse
Total Timeline: 6-8 weeks for sea freight with proper documentation
Step 5: Understanding Costs Beyond Product Price
The Real Cost Breakdown:
Product Cost: 100%
Sea Freight: +15-25% (depending on volume)
Insurance: +1-2%
Customs Duty: Variable (0-20% depending on product/market)
Testing/Certification (if needed): +5-10%
Customs Broker Fees: +$200-500 per shipment
Storage/Warehousing: Variable
Example: $10,000 product order might total $13,000-15,000 landed cost
Step 6: Red Flags to Watch For
Warning signs of unreliable suppliers:
❌ Can't provide GMP certificate or CoA
❌ Vague about ingredient sourcing
❌ Won't share lab testing reports
❌ Promises impossibly fast timelines
❌ Avoids questions about compliance
❌ No experience with your target market
❌ Payment terms that feel risky
Step 7: Making Your First Import Smooth
Best Practices from Experienced Buyers:
Start Small
- Order smaller quantities initially (500-1000 units)
- Test product quality before scaling
- Verify documentation accuracy
- Build relationship before major commitment
Use Established Trade Routes
- Major ports (Mumbai, Chennai) have smoother processes
- Avoid experimental shipping methods
- Work with freight forwarders experienced in wellness products
Build Buffer Time
- Add 2-3 weeks to quoted timelines for first orders
- Account for potential customs inspections
- Plan inventory accordingly
Communicate Clearly
- Specify exact market requirements upfront
- Share label compliance needs early
- Confirm testing standards match your market
- Get everything in writing
Step 8: Long-Term Partnership Considerations
Questions to Ask Potential Suppliers:
About Capacity:
- What's your minimum order quantity?
- Can you scale if my business grows?
- What's lead time for reorders?
About Quality:
- How do you ensure batch consistency?
- What happens if a batch fails testing?
- Can I visit your facility?
About Support:
- Do you help with market-specific compliance?
- Can you adjust formulations for regulations?
- What documentation support do you provide?
The Bottom Line
Importing Ayurvedic products from India is a proven path for wellness brands worldwide. The key is working with suppliers who understand that documentation isn't bureaucracy—it's how you protect your customers, your brand, and your business.
With the right partner, you get:
- Clear documentation from day one
- Products formulated for your market
- Transparent communication throughout
- Support when regulatory questions arise
The complexity isn't in the process itself—it's in finding a supplier who treats your market compliance as seriously as you do.